Reporting on finance and banking news in the Middle East
Provided by AGPGlobal Stem Cell Therapy Market Set to Flourish at a CAGR of ~11% by 2034 Owing to the Technological Advancements and Favorable Regulations | DelveInsight
The stem cell therapy market is witnessing strong growth driven by rising demand for regenerative medicine and advanced treatment options for chronic diseases such as cancer, diabetes, and neurological disorders. Increasing investments in research, clinical trials, and biotechnology innovation are accelerating the development of new stem cell-based therapies. Supportive regulatory frameworks and growing adoption of personalized medicine are further boosting market expansion.
New York, USA, May 05, 2026 (GLOBE NEWSWIRE) -- Global Stem Cell Therapy Market Set to Flourish at a CAGR of ~11% by 2034 Owing to the Technological Advancements and Favorable Regulations | DelveInsight
The stem cell therapy market is witnessing strong growth driven by rising demand for regenerative medicine and advanced treatment options for chronic diseases such as cancer, diabetes, and neurological disorders. Increasing investments in research, clinical trials, and biotechnology innovation are accelerating the development of new stem cell-based therapies. Supportive regulatory frameworks and growing adoption of personalized medicine are further boosting market expansion.
DelveInsight’s Stem Cell Therapy Market Insights report provides the current and forecast market analysis, individual leading stem cell therapy companies’ market shares, challenges, stem cell therapy market drivers, barriers, trends, and key stem cell therapy companies in the market.
Stem Cell Therapy Market Summary
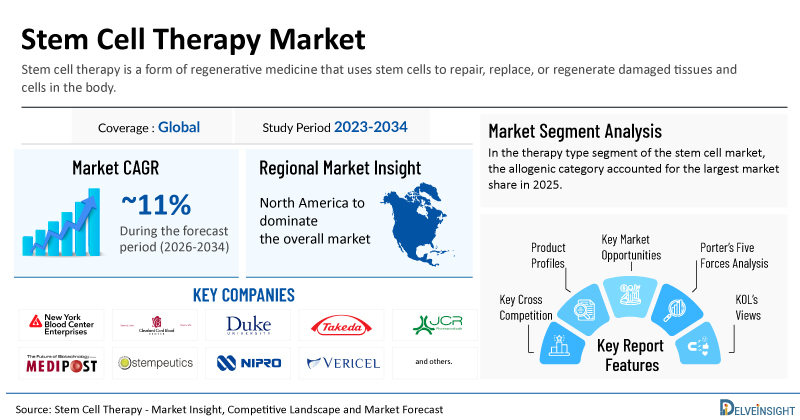
- 2025 Stem Cell Therapy Market Size: USD 21.9 Billion
- 2034 Projected Stem Cell Therapy Market Size: USD 58.5 Billion
- Stem Cell Therapy Market Growth Rate (2026-2034): ~11%
- Largest Stem Cell Therapy Market: North America
- Largest Therapy Type Segment: Allogenic Category
- Key Companies in the Stem Cell Therapy Market: New York Blood Center, Cleveland Cord Blood Center, Duke University, Takeda Pharmaceutical Company, JCR Pharmaceuticals, Medipost, Stempeutics Research, Nipro Corporation, Vericel Corporation, Holostem Terapie Avanzate, Orchard Therapeutics, Bluebird Bio, Cynata Therapeutics, Sumitomo Pharma Co., Ltd., and others
To read more about the latest highlights related to the stem cell therapy market, get a snapshot of the key highlights @ https://www.delveinsight.com/sample-request/stem-cell-market
Key Factors Contributing to the Rise in Growth of the Stem Cell Therapy Market
- Increasing Prevalence of Chronic and Degenerative Diseases: Rising cases of conditions such as cancer, diabetes, cardiovascular disorders, neurological diseases, and autoimmune disorders are driving demand for regenerative treatment options like stem cell therapy.
- Advancements in Regenerative Medicine Research: Continuous progress in stem cell biology, gene editing, tissue engineering, and cell expansion technologies has improved the safety, efficacy, and scalability of stem cell therapies.
- Growing Clinical Trial Activity: A significant increase in clinical studies evaluating stem cell-based treatments across multiple therapeutic areas is accelerating product development and market confidence.
- Supportive Regulatory Frameworks: Favorable government initiatives, expedited approval pathways, and increasing regulatory support for regenerative medicines in countries such as Japan, the United States, and South Korea are boosting commercialization.
- Rising Investments and Funding: Increased investments from biotech companies, pharmaceutical firms, venture capitalists, and public organizations are supporting innovation and manufacturing expansion.
- Growing Demand for Personalized Medicine: Stem cell therapies offer patient-specific treatment approaches, making them attractive in precision medicine and targeted healthcare strategies.
- Expanding Applications Across Therapeutic Areas: Beyond oncology, stem cell therapies are gaining traction in orthopedics, dermatology, ophthalmology, cardiology, and neurology, broadening market opportunities.
- Improved Manufacturing and Storage Infrastructure: Advances in cell banking, cryopreservation, and large-scale manufacturing processes are making stem cell products more accessible and commercially viable.
- Rising Awareness and Acceptance: Increasing awareness among healthcare professionals and patients regarding the potential of stem cell therapies is supporting adoption globally.
Get a sneak peek at the stem cell therapy market dynamics @ Stem Cell Therapy Market Trends
Regional Stem Cell Therapy Market Insights
North America
- Among all regions, North America held the largest share of the stem cell market in 2025, accounting for 48.87%.
- The region is anticipated to lead the market owing to its advanced healthcare infrastructure, substantial investments in regenerative medicine, and a significant number of ongoing clinical trials and product approvals.
- Moreover, the rising burden of chronic diseases, strong presence of renowned research institutions, and leading industry participants further support the early adoption and commercialization of innovative stem cell therapies across the region.
Europe
- Europe’s stem cell market is witnessing significant expansion, supported by favorable regulatory policies, rising public and private funding, and ongoing progress in regenerative medicine.
- The region benefits from a structured regulatory pathway established by the European Medicines Agency for Advanced Therapy Medicinal Products (ATMPs), enabling faster development and market entry of stem cell therapies.
- In addition, growing partnerships among academic institutions, biotechnology firms, and government organizations are encouraging innovation and strengthening the pipeline of stem cell-based treatments across areas such as ophthalmology, orthopedics, and rare diseases.
- Together, these factors, including regulatory support, sustained R&D investments, key product approvals, and an increasing disease burden, are strongly contributing to the growth of the stem cell market in Europe.
Asia-Pacific
- The Asia Pacific region is becoming a key engine of growth for the Stem Cell Research market, supported by favorable government initiatives, faster clinical adoption, growing biotechnology capabilities, increasing healthcare spending, and a vast patient base with significant unmet medical needs.
- Nations including China, Japan, South Korea, and India are leading efforts in advancing both stem cell research and commercial development.
- Japan has especially strengthened its position through an accelerated approval pathway for regenerative medicine, driving major milestones such as the March 2026 conditional approval of iPSC-based therapies like ReHeart and Amusepri for heart failure and Parkinson’s disease, representing some of the first real-world uses of reprogrammed stem cell treatments globally.
To know more about why North America is leading the market growth in the stem cell therapy market, get a snapshot of the Stem Cell Therapy Market Share
Recent Developmental Activities in the Stem Cell Therapy Market
- In March 2026, Japan approved Amchepry, the world’s first regenerative medicine treatment using induced pluripotent stem (iPS) cells to treat Parkinson’s disease.
- In December 2025, Cipla Limited announced the launch of Ciplostem, an innovative allogeneic mesenchymal stromal cell (MSC) therapy for Knee Osteoarthritis (Knee OA), approved by the Drug Controller General of India (DCGI). Developed by Stempeutics Research, the therapy offers a disease-modifying treatment option targeting Grade II and III Knee OA and marks a significant advancement in Cipla’s entry into Orthobiologic medicine.
- In April 2025, an off-the-shelf, iPSC-derived CAR T-cell therapy for the treatment of active moderate to severe systemic lupus erythematosus (SLE), including lupus nephritis (LN), was granted an FDA RMAT designation for entering Phase I clinical trial.
- In February 2025, the FDA granted IND clearance for Fertilo (Figure 2), the first iPSC-based therapy to enter a U.S. Phase III trial.
What is Stem Cell Therapy?
Stem cell therapy is a form of regenerative medicine that uses stem cells to repair, replace, or regenerate damaged tissues and cells in the body. Stem cells are unique because they have the ability to develop into different specialized cell types, such as muscle cells, nerve cells, or blood cells, depending on the body’s needs. This therapy is being explored and used for treating a range of conditions, including blood disorders, certain cancers, orthopedic injuries, neurological diseases, and autoimmune disorders. Common sources of stem cells include bone marrow, adipose tissue, umbilical cord blood, and laboratory-grown pluripotent stem cells. Stem cell therapy aims to restore normal function, promote healing, and improve patient outcomes, making it a promising area of modern healthcare and biomedical research.
| Stem Cell Therapy Market Report Metrics | Details |
| Coverage | Global |
| Study Period | 2023–2034 |
| Stem Cell Therapy Market CAGR | ~12% |
| Stem Cell Therapy Market Size by 2034 | USD 58.5 Billion |
| Key Stem Cell Therapy Companies | New York Blood Center, Cleveland Cord Blood Center, Duke University, Takeda Pharmaceutical Company, JCR Pharmaceuticals, Medipost, Stempeutics Research, Nipro Corporation, Vericel Corporation, Holostem Terapie Avanzate, Orchard Therapeutics, Bluebird Bio, Cynata Therapeutics, Sumitomo Pharma Co., Ltd., and others |
Stem Cell Therapy Market Assessment
-
Stem Cell Therapy Market Segmentation
- Stem Cell Therapy Market Segmentation By Therapy Type: Allogenic and Autologous
- Stem Cell Therapy Market Segmentation By Cell Source: Adult Stem Cells {Hematopoietic Stem Cells (HSCs), Mesenchymal Stem Cells (MSCs), and Others}, Embryonic Stem Cells (ESCs), and Induced Pluripotent Stem Cells (iPSCs)
- Stem Cell Therapy Market Segmentation By Therapeutic Application: Hematological Disorders, Musculoskeletal Disorders, Neurological Disorders, and Others
- Stem Cell Therapy Market Segmentation By Technology: Cell Collection & Isolation, Cell Expansion & Culturing, Cryopreservation & Storage, and Others
- Stem Cell Therapy Market Segmentation By End User: Hospitals & Transplant Centers, Academic & Research Institutes, and Others
- Stem Cell Therapy Market Segmentation By Geography: North America, Europe, Asia-Pacific, and Rest of World
- Porter’s Five Forces Analysis, Product Profiles, Case Studies, KOL’s Views, Analyst’s View
Which MedTech key players in the stem cell therapy market are set to emerge as the trendsetter explore @ Stem Cell Therapy Market Analysis
Table of Contents
| 1 | Stem Cell Therapy Market Report Introduction |
| 2 | Stem Cell Therapy Market Executive Summary |
| 3 | Stem Cell Therapy Market Key Factors Analysis |
| 4 | Impact Analysis |
| 5 | Regulatory Analysis |
| 6 | Stem Cell Therapy Market Porter’s Five Forces Analysis |
| 7 | Stem Cell Therapy Market Assessment |
| 8 | Competitive Landscape |
| 9 | Startup Funding & Investment Trends |
| 10 | Stem Cell Therapy Market Company and Product Profiles |
| 11 | KOL Views |
| 12 | Project Approach |
| 13 | About DelveInsight |
| 14 | Disclaimer & Contact Us |
Interested in knowing the stem cell therapy market share by 2034? Click to get a snapshot of the Stem Cell Therapy Market Size
Related Reports
Hematopoietic Stem Cell Transplantation Market
Hematopoietic Stem Cell Transplantation Market Insights, Epidemiology, and Market Forecast – 2036 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key HSCT companies including Actinium Pharmaceuticals, Medexus Pharmaceuticals, medac Pharma, BioLineRx, Sanofi, and others.
Hematopoietic Stem Cell Transplantation Clinical Trial Analysis
Hematopoietic Stem Cell Transplantation Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key HSCT companies, including Actinium Pharmaceuticals, BioLineRx, Athersys, Novartis, CareDex, Orchard Therapeutics, Magenta Therapeutics, Graphite Bio, Vor Biopharma, Jasper Therapeutics, Garuda Therapeutics, and others.
Allogeneic Hematopoietic Stem Cell Transplant Market
Allogeneic Hematopoietic Stem Cell Transplant Market Insights, Epidemiology, and Market Forecast – 2036 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key HSCT companies including Merck Sharp & Dohme LLC, In8bio Inc., Incyte Corporation, Forge Biologics, Inc, GlaxoSmithKline, Bellicum Pharmaceuticals, SecuraBio, ModernaTX, Inc., Miltenyi Biotec, Inc., Jazz Pharmaceuticals, Gilead Sciences, Celularity Incorporated, Seres Therapeutics, Inc., Omeros Corporation, Novartis, Marker Therapeutics, Inc., and others.
Multiple Myeloma Market Insights, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key multiple myeloma companies, including Johnson & Johnson (Janssen), Pfizer, AbbVie and Roche (Genentech), Regeneron Pharmaceuticals, Bristol-Myers Squibb, Celgene, Roche (Genentech), Arcellx, Novartis, Regeneron Pharmaceuticals, BeiGene, CARsgen Therapeutics, Cartesian Therapeutics, C4 Therapeutics, Heidelberg Pharma, Bristol-Myers Squibb, RAPA Therapeutics, AbbVie (TeneoOne), Takeda, and others.
Non-Hodgkin’s Lymphoma Market Insights, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key NHL companies, including Novartis, AstraZeneca, Genentech, BioInvent, Genmab, SystImmune, Nordic Nanovector, Pacylex Pharmaceuticals, Artiva Biotherapeutics, Inc., Chipscreen Biosciences, Ltd., Timmune Biotech Inc., Chia Tai Tianqing Pharmaceutical Group Co., Ltd., Gilead Sciences, Acerta Pharma BV, Adagene Inc, Conjupro Biotherapeutics, Inc., Rhizen Pharmaceuticals, Juventas Cell Therapy Ltd., Incyte Corporation, HUYA Bioscience International, SecuraBio, Genor Biopharma Co., Ltd., Kyowa Kirin Co., Ltd., Antengene Therapeutics Limited, Regeneron Pharmaceuticals, Jiangsu HengRui Medicine Co., Ltd., Xynomic Pharmaceuticals, Inc., BioTheryX, Inc., UWELL Biopharma, Kronos Bio, Bio-Thera Solutions, Spectrum Pharmaceuticals, Inc., Aptose Biosciences Inc., Miltenyi Biomedicine GmbH, Precision BioSciences, Inc., Teneobio, Inc., TCR2 Therapeutics, IGM Biosciences, Inc, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.

Contact Us Shruti Thakur info@delveinsight.com +14699457679
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
